Equation Tin React With Acidified Potassium Permanganate
Using the reaction to test for carbon-carbon double bonds. So in terms Of loss gain in oxidation number Mn in acidified potassium manganate VII decreased from 7 to 2 while that of sulphur in sulphur dioxide increased from 4 to 6.

Oxidation Of Alcohols By Potassium Permanganate The Mechanism Chemistry Education Chemistry Basics Organic Chemistry
If the combustible material is.
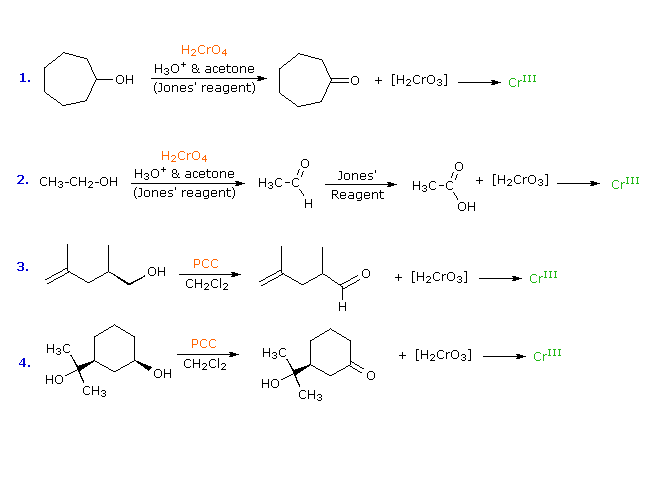
Equation tin react with acidified potassium permanganate. Equation One MnO4â 8H. The equation for the oxidation of oxalate by permanganate in acidic solution is 2 MnO 4 - aq 5 H 2 C 2 O 4 aq 6 H 3 O aq -- 2Mn 2 aq 10 CO 2 aq 14 H 2 O Demonstration Notes Warnings Safety Information etc. Concepts CoveredReduction and Oxidation in reactionsTransfer of Ions in reactionsGrade 8- Chemical Reactions and their TypesGrade 10- Chemical Reactions a.
Sulfur dioxide gas SO2 is bubbled through a solution of acidified potassium permanganate KMnO4. __ Sn2 __MnO4- __ H __ Sn4 __Mn2 __ H2O a Identify the oxidizing agent and the reducing agent. When an acidic solution of tinII chloride is added to a solution of potassium permanganate the purple color rapidly fades.
- About 10 cm. Noncombustible but accelerates the burning of combustible material. Nitric acid - diluted solution.
CID 422689 Permanganic acid Date s. Begingroup Permanganic acid ceHMnO4 is a strong acid so its not easy to form via protonation of ceMnO4-. The overall reaction takes place in the process is.
Tin II ions can be oxidized to tin IV ions by acidified potassium permanganate VII solution according to the following unbalanced equation. The unbalanced equation for this reaction is. Oxidation reaction Cl- rarr 12Cl_2 e- i Reduction reaction MnO_4- 8H 5e- rarr Mn2 4H_2O ii In each reaction both charge and mass are balanced are they.
An oxidation-reduction reaction occurs. __ Sn2 __MnO4 - __ H __ Sn4 __Mn2 __ H2O Can you please explain to me how to balance this equation. The unbalanced equation for this reaction is.
Equation Two SO2 4H 2e SO42 2H2O. Reduction Half reaction- MnO 4 8H 5e Mn 2 4H 2 O x 2. Avoid contact with skin and eyes.
Tin II chloride react with potassium permanganate and hydrogen chloride to produce hydrogen hexachloridostannate IV manganese II chloride potassium chloride and water. They can oxidize Fe2 ions to Fe3 ions. TinII ions can be oxidized to tinIV ions by acidified potassium.
PermanganateVII solution according to the following unbalanced equation. Molarity Fe II 5x molarity MnO4 volume MnO4 volume Fe II 2. Equation One MnO4 8H 5e Mn2 4H2O.
Write the chemical equation for the oxidation of cyclohexene with acidified K M n O 4. Ii Nitrogen is obtained in the reaction of aqueous ammonia with potassium permanganate. Sulfur dioxide gas SO2 is bubbled through a solution of acidified potassium permanganate KMnO4.
Potassium permanganate appears as a purplish colored crystalline solid. In the specific case I mentioned. KMnO_4 4H_2SO_4 5KCl rarr MnSO_4 52Cl_2 3K_2SO_4 4H_2O This is a redox reaction.
Dihydroxylation KMnO4 Dihydroxylation Definition. MnO4 SO2 Mn2 SO42. When treated alkene with potassium permanganate two hydroxyl groups are added so the resulting compounds are called dihydroxylates of the reaction of syn-dihydroxylation.
MnO4 SO2 Mn2 SO42. Constructing equations for redox reactions Method 1. Sn 2 2 Fe 3 Sn 4 2 Fe 2.
Hence SO2 is oxidised whereas potassium permanganate is reduced. Oxidation Half reaction- C 2 O 4 2- 2CO 2 2e x 5. A student wrote the two following half-equations.
Write balanced equation for the follownig i Potassium permanganate is added to hot solution of manganous sulphate. Acidified manganateVII ions are a powerful oxidizing agent. But equally it could be any one of a large number of other compounds all of which can be oxidised by manganateVII ions under.
Equation Two SO2 4H 2e SO42 2H2O. At the same time the manganateVII ions are reduced to Mn2 ions. Combining half-equations which show numbers of electrons transferred Example.
Of approximately 1 mol dm 3. Hydrogen chloride - concentrated solution. The overall reaction is.
MnO4â SO2 Mn2 SO42â A student wrote the two following half-equations. The purple colour disappears and a brown precipitate is formed. If an organic compound reacts with dilute alkaline potassium manganateVII solution in the cold to give a green solution followed by a dark brown precipitate then it may contain a carbon-carbon double bond.
An oxidation-reduction reaction occurs. MnO 4 8 H 5 Fe 2 2 Mn 5 Fe 3 4 H 2O To ensure that all of the iron in the unknown is in the Fe 2 state prior to titration the unknown is treated with tinII chloride which reduces any small amount of Fe 3 that might be present. Addition of tinII chloride results in a colorless solution.
Overall reaction- 2KMnO 4 3H 2 SO 4 5COOH 2 K 2 SO 4 2MnSO 4 8H 2 O 10CO 2 The ionic equation involved in the process is given below. Knowing the molarity of your KMnO4 and the volume used in each titration you can calculate the molarity of your Fe II solution as. 5SnCl 2 2KMnO 4 26HCl 5H 2 SnCl 6 2MnCl 2 2KCl 8H 2 O.
Sulfur dioxide gas SO2 is bubbled through a solution of acidified potassium permanganate KMnO4. Equation One MnO4 8H 5e Mn2 4H2O. A student wrote the two following half-equations.
- About 1 g of powdered tin optional. Check the balance Sodium nitrite react with potassium permanganate and sulfuric acid to produce sodium nitrate manganese II sulfate potassium sulfate and water. Dissolve 4 g of potassium permanganate in 100 cm3 of 1 mol dm 3 sulfuric acid.
An oxidation-reduction reaction occurs. And the equations must. 5NaNO 2 2KMnO 4 3H 2 SO 4 5NaNO 3 2MnSO 4 K 2 SO 4 3H 2 O.
Permanganate ion is reduced to Mn2 and chloride ion is oxidized to chlorine gas. 3 potassium permanganate potassium manganateVII KMnO4 in 1 mol dm3 sulfuric acid. The chemical equation is.
The unbalanced equation for this reaction is. When adding permanganate ions to very very acidic mixtures for example pure liquid ceH2SO4 you can consider that the reaction ceMnO4- H HMnO4 happens to an appreciable extent producing permanganic acid. Reaction with Potassium Permanganate.
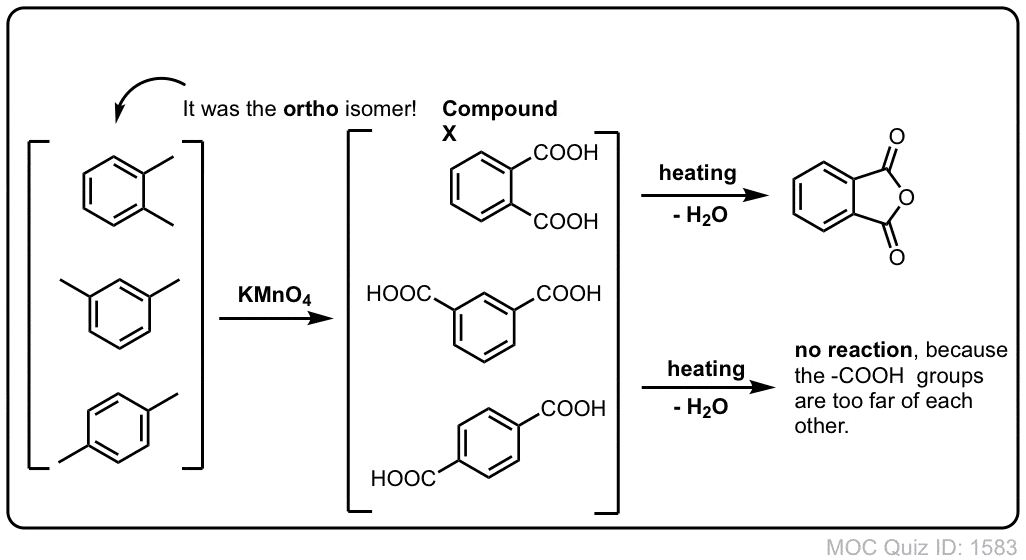
Oxidation Of Aromatic Alkanes With Kmno4 To Give Carboxylic Acids Master Organic Chemistry
Comments
Post a Comment