Mangantae With Tin Ion
Environmental Protection Agency has also set a Health Advisory for manganese of 03 mgL. Using the same stages as before start by writing down what you know.

Redox Titrations Video Khan Academy
66625 Adapted Stein Brown method Melting Pt deg C.

Mangantae with tin ion. It is a manganese oxoanion and a divalent inorganic anion. The oxidising agent is the dichromateVI ion Cr 2 O 7 2-. Then change the name according to the following table.
Tin modified sodium manganese hexacyanoferrate as a Prussian blue analogue is studied as a cathode material for sodium ion batteries. The simplest ion that manganese forms in solution is the hexaaquamanganeseII ion - MnH 2 O 6 2. MnO 4 - 8H 5 e - Mn 2 4H 2 O.
Sn tin stannum Sn 2 tinII ion stannous ion Sn 4 tinIV ion stannic ion Pb lead plumbum Pb 2 leadII ion plumbous ion Pb 4 leadIV ion plumbic ion Sb antimony stibium Sb 3 antimonyIII ion antimonous ion Sb 5 antimonyV ion antimonic ion Bi bismuth Bi 3. It is a conjugate base of a hydrogen manganate. Fit the tube with a rubber bung.
- About 1 g of powdered tin optional. Indeed manganates are often isostructural with sulfates and chromates a fact first noted by Eilhard Mitscherlich in 1831. By co-precipitation of Sn 4 during the synthesis process the modified sodium manganese hexacyanoferrate materials crystallize with face-centered cubic structure with space group Fm 3 m while the unmodified one possesses a.
This time you would need to multiply the iron half-reaction by 5 in order to produce the 5 electrons needed by the second half-reaction. If you arent happy about complex ions including the way they are bonded and named it would pay you to follow this link and explore the first couple of pages in the complex ions menu before you go on. 29026 Mean or Weighted MP VPmm Hg25 deg C.
If the combustible material is. The E set involves the x2 - y2 and z2 orbitals and the T2 set involves the other three. Manganate is a divalent inorganic anion obtained by removal of both protons from manganic acid.
Mn 2 aq H 2 O 2 aq 2 OH aq MnO 2 s brown-black 2 H 2 O l The general rule for manganese compounds reacting with a surplus of hydrogen peroxide under acidic conditions is the oxidation to Mn II. A permanganate is the general name for a chemical compound containing the manganateVII ion MnO 4Because manganese is in the 7 oxidation state the permanganateVII ion is a strong oxidizing agentThe ion has tetrahedral geometry. The manganateVI ion is tetrahedral similar to sulfate or chromate.
Permanganate is a manganese oxoanion and a monovalent inorganic anion. - About 10 cm. All of the formulas for acids that are dealt with in this section will start with the hydrogen ion.
The manganeseoxygen distance is 1659 pm about 3 pm longer than in permanganate. The formula for Tin Oxide is SnO2 depending on the assigned charge for tin. Anions 1-acetate C 2 H 3 O 2-cyanide CN-amide NH 2.
Analysis of Manganate Permanganate Anion Standards and Calendar Aged Anode Samples by IC PO71671-EN 0515S Analysis and Identification of Ionic Manganese Containing Degradation Products from the Anode Surface of Cycle Calendar Aged Li-Ion Batteries Paul Voelker1 Kate Comstock2 Rosanne Slingsby1 Charanjit Saini1 and Chris Pohl1. The T2 set is raised in energy and the E set is lowered in energy. For these reasons it is recommended that drinking water have no more than 03 mgL or 03 parts per million of iron and less than 005 mgL of manganese.
Boiling Pt deg C. Using a small measuring cylinder add 80 cm3 of the acidified ethanedioic acid solution to the test tube. The hydrogen ion in acidified potassium manganate VII 2.
In the presence of a tetrahedral ligand field the metal d ions split into a E set and a T2 set. The splitting is particularly low O2- is a pi donor ligand and its a tetrahedral. In order to name the compound as an acid first name the compound as if it were an ionic compound.
What happens to potassium if potassium permanganate is reduced in a redox reaction. Redox Reaction Equation with elusive oxygen. Zero a stop clock ready for use.
Predicted data is generated using the US Environmental Protection Agencys EPISuite. Noncombustible but accelerate the burning of combustible material. Mn II-ions are readily oxidized to MnO 2 by hydrogen peroxide under alkaline conditions.
Redox reaction with tin and sulfuric acid solution. Of approximately 1 mol dm 3. Then add them together to give c The reaction between ethanol and acidified potassium dichromateVI solution to give.
Balance the oxygens by adding a water molecule to the left-hand side. IronIII a 3 charge. Permanganate ion reduces to a manganese II ion in the acidic solution.
B The reaction between ironII ions and acidified potassium manganateVII solution. The exact chemical reaction is dependent upon the organic. In this case the charge assigned to tin was 4.
Manganese often results in a dense black stain or solid. The formula for the tin four ion is Sn4. In this experiment you will use a standard solution of potassium permanganate KMnO 4 to determine the of iron as Fe 2 in an unknown solution.
This is reduced to chromiumIII ions Cr 3. Table for Naming Acids. Well do the ethanol to ethanoic acid half-equation first.
It is a conjugate base of a permanganic acid. 4Sn tinIV ion stannic ion Lead Pb2 leadII ion plumbous ion 4Pb leadIV ion plumbic ion Bismuth Bi3 bismuthIII ion Bi5 bismuthV ion Main-Group Nonmetals Groups IVA VA VIA and VIIA Group IVA VA VIA and VIIA nonmetals tend to form anions by gaining enough electrons to fill their valence shell with eight electrons. The new term single-ion magnet SIM was employed though the magnetic properties including the bistability were correlated to the molecular structure in particular the highly uniaxial coordination environment dictated by the staggered configuration of the phthalocyaninate rings in the double-decker structure see Fig.
Log Octanol-Water Partition Coef SRC. Permanganate solutions are purple in color and are stable in neutral or slightly alkaline media. Log Kow KOWWIN v167 estimate 193 Boiling Pt Melting Pt Vapor Pressure Estimations MPBPWIN v142.
TinIV Sn4 nickelIV Ni4 leadIV Pb4 Roman numeral notation indicates charge of ion when element commonly forms more than one ion. Place 20 cm3 of a 0002 mol dm-3 solution of potassium manganateVII in a test tube. Dissolve 4 g of potassium permanganate in 100 cm3 of 1 mol dm 3 sulfuric acid.
Use hydrogen as the cation. Reaction of manganese with peroxide. 3 potassium permanganate potassium manganateVII KMnO4 in 1 mol dm3 sulfuric acid.
As a d 1 ion it is paramagnetic but any JahnTeller distortion is too small to be detected by X-ray crystallography. For example ironII has a 2 charge. This reaction requires 5 electrons and 8 hydrogen ions.
Reduction Of The Metal Centre In Aqueous Permanganate Ion Involves
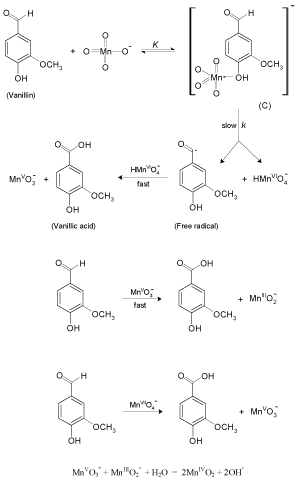
Kinetics And Mechanism Of Oxidation Of Vanillin By Permanganate In Neutral Medium And The Effect Of Different Transition Metal Ion Catalysts

Comments
Post a Comment