Tin Oxide + Carbon Equation
This means that some metals can be extracted from their metal oxides using carbon. The general equation for the reactions in which metal oxides are reduced by heating with carbon.

Reactivity Series Google Search Chemistry Worksheets Chemical Reactions Covalent Bonding
The equation can be written as.

Tin oxide + carbon equation. The highest faradaic and energy efficiencies of 64 and 27 were obtained at 140 V vs. It is a colourless diamagnetic amphoteric solid. The equation for the event of photosynthesis iswater carbon dioxide --using sunlight-- energy oxygen.
00739 moles of tin IV oxide reacts with carbon according to the reaction equation shown below. Write and balance the equation. Then 4 x 0065moles 0261moles of C.
It is this target producing liquid teen plus guy Rundle knocks I we need to interpret this reaction in terms of particles moles on must. Metal oxide carbon metal carbon monoxide. The most prominent example is the production of iron from iron oxide in a blast furnace.
What is the word equation for the reaction between lead oxide and carbon. For tin oxide the reduction with carbon or coke is an established industrial process. Metal is reduced from a positive oxidation state in the metal oxide to the metallic element.
So 14g2147726gmole 0065mole of SnO4. Quite resistant minimum reaction Carbon Oxygen --. The electrical properties of tin iv oxide render it an excellent material for the detection of very low levels of carbon monoxide and several devices based on sintered pellets of SnO.
SnO 2 2 H 2 SO 4 SnSO 4 2 2 H 2 O. So well start then with our 283 g carbon dioxide converted into moles carbon dioxide by dividing by the molar mass of carbon dioxide 441 g per mole. What is the balanced equation of the decomposition of.
Our mission is to. Scheme shows the interaction of tin oxide with graphitic carbon nitride which resulted in a coral-shaped structure shown in SEM image. It also shows the interaction of tin oxide and graphitic carbon nitride with H2O2 in the presence of TMB 33 55-Tetramethylbenzidine and as a consequence TMB gets oxidized and ended up in change in color.
However for some metals this fails and metal carbides are formed instead. Metal oxide carbon metal carbon dioxide. Carbon monoxide is usually treated as if it was a neutral oxide but in fact it is very very slightly acidic.
A comparative study is reported on the electrocatalytic reduction of CO 2 to HCOOH in aqueous alkaline solution with differently prepared tin-oxide particles on multi-walled carbon nanotubes from SnCl 2 or SnCl 4 precursors. Reaction of Iron III oxide Fe2O3 with carbon monoxide CO to form iron Fe and carbon dioxide CO2 is a redox reaction as both oxidation of carbon monoxide and reduction of iron is taking place. Find step-by-step Chemistry solutions and your answer to the following textbook question.
Blank SnO2 blank C arrow blank Sn. TinII oxide stannous oxide is a compound with the formula SnOIt is composed of tin and oxygen where tin has the oxidation state of 2. This works for zinc iron tin lead and copper.
SnO 2 2NaOH Na 2 SnO 3 H 2 O. SnO4tin IV 4Cs Sns 4COg Determine ratio 1 SnO4. Tin was among the first metals used by humans.
Tin oxide also known as stannic oxide is the inorganic compound with the formula SnO2. This works for zinc iron tin lead and copper. The products of this reaction are tin and carbon dioxide.
The chemical equation is given below. When tinIV oxide is heated with carbon in a process called smelting the element tin can be extracted mathrmSnO_2mathrms2 mathrmCmathrms rightarrow mathrmSnl2 mathrmCOmathrmg Interpret the chemical equation in terms of particles moles and mass. Carbon and silicon oxides.
CO2 youll get gas which you might might not want to collect ie if you want to collect tin only and dont mind. There are two forms a. Copper is the least reactive of these five metals.
Copper is the least reactive of these five metals. With many other names this oxide of tin is an important material in tin chemistry. Carbon at first is partially oxidised to form carbon monoxide PbO C Pb CO which then is involved in further reduction PbO.
SCE with particles that were obtained by KBH 4 reduction from a SnCl 2. Can lead be extracted using carbon. What happens when lead oxide is heated with carbon.
This reaction This equation is saying that one unit often for oxide reacts with two atoms both carbon again. Tin oxides such as SnO 2 SnO and mixed-valence oxides Sn 2 O 3 or Sn 3 O 4 have been widely investigated due to their exceptional semiconducting properties which make them promising candidates for many modern applications. Tin oxygen --.
Metal oxide carbon metal carbon dioxide. The chemical equation is given below. Then when we have moles of carbon dioxide we can convert into moles of what were interested in.
Carbon fits between aluminium and zinc in the. Carbon is a non-metal but it is more reactive than some metals. The mineral form of SnO2 is called cassiterite and this is the main ore of tin.
When tinIV oxide reacts with sodium hydroxide stannate IV sodium and water are formed. Magnesium carbonate with a 1 to 1 relationship and we get 643 moles of magnesium carbonate will be required to produce 283 g carbon dioxide. 00949 moles of tin IV oxide reacts with carbon according to the reaction equation shown below.
Elemental tin is produced by heating tinIV oxide the principal ore of tin with carbon. After 15 minutes small globules of molten lead were produced. SnO2 C Sn CO Calculate the number of moles of tin that are produced if all 00739 moles of tin IV oxide are consumed during the reaction.
First they do the interpretation in terms of particles. At a temperature of 350-400C this reaction takes place. An oxide which can show both acidic and basic properties is said to be amphoteric.
In turn carbon is oxidized to carbon monoxide and carbon dioxide. 1 For instance tin dioxide SnO 2 a wide band gap 36 eV n-type semiconductor with high electronic conductivity and high optical transparency in the visible range is. Tin IV oxide reacts with carbon to form tin and carbon monoxide.
The trend is therefore from acidic oxides at the top of the Group towards amphoteric ones at the bottom. Write the coefficients to balance the chemical equation for the following reaction. Uses of Tin Oxide SnO 2.
SnO2 C Sn CO Calculate the number of moles of tin that are produced if all 00949 moles of tin IV oxide are consumed during the reaction. Given the violence equation but the reaction between teen for oxide and carbon this is soul.
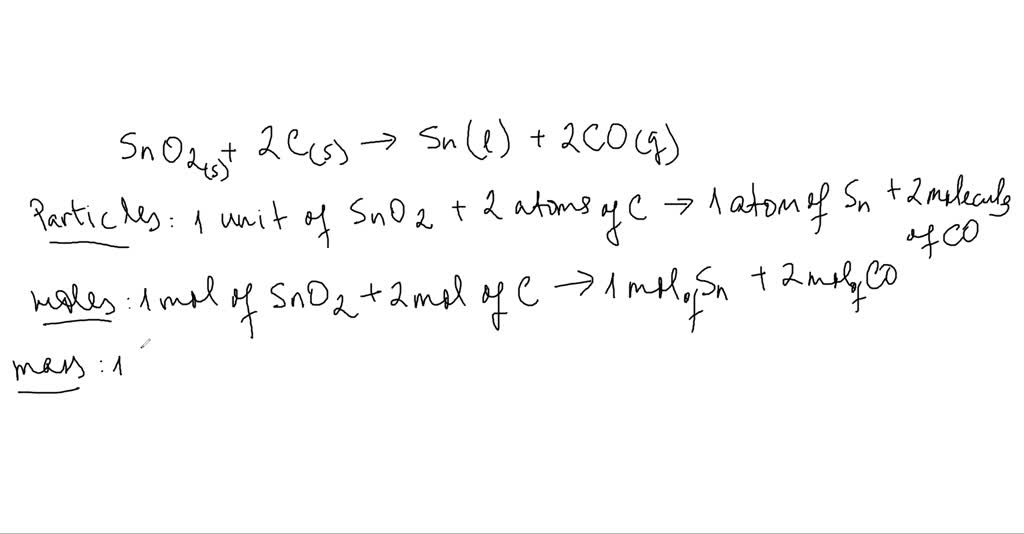
Solved Smelting When Tin Iv Oxide Is Heated With Carbon In A Process Called Smelting The Element Tin Can Be Extracted Begin Equation Mathrm Sno 2 Mathrm S 2 Mathrm C Mathrm S Rightarrow Mathrm Sn 1 2 Mathrm Co Mathrm G End
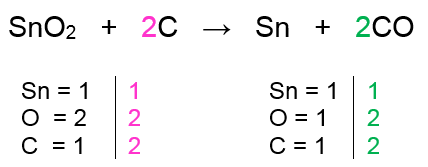
0 0949 Moles Of Tin Iv Oxide Reacts With Carbon According Clutch Prep
Comments
Post a Comment