A Tin Is Shrinking After Boiled Boyles Law
For this you can use the stove or a heater. As per Boyles law when a pressurized gas enters a vacuum region it will expand infinitely.
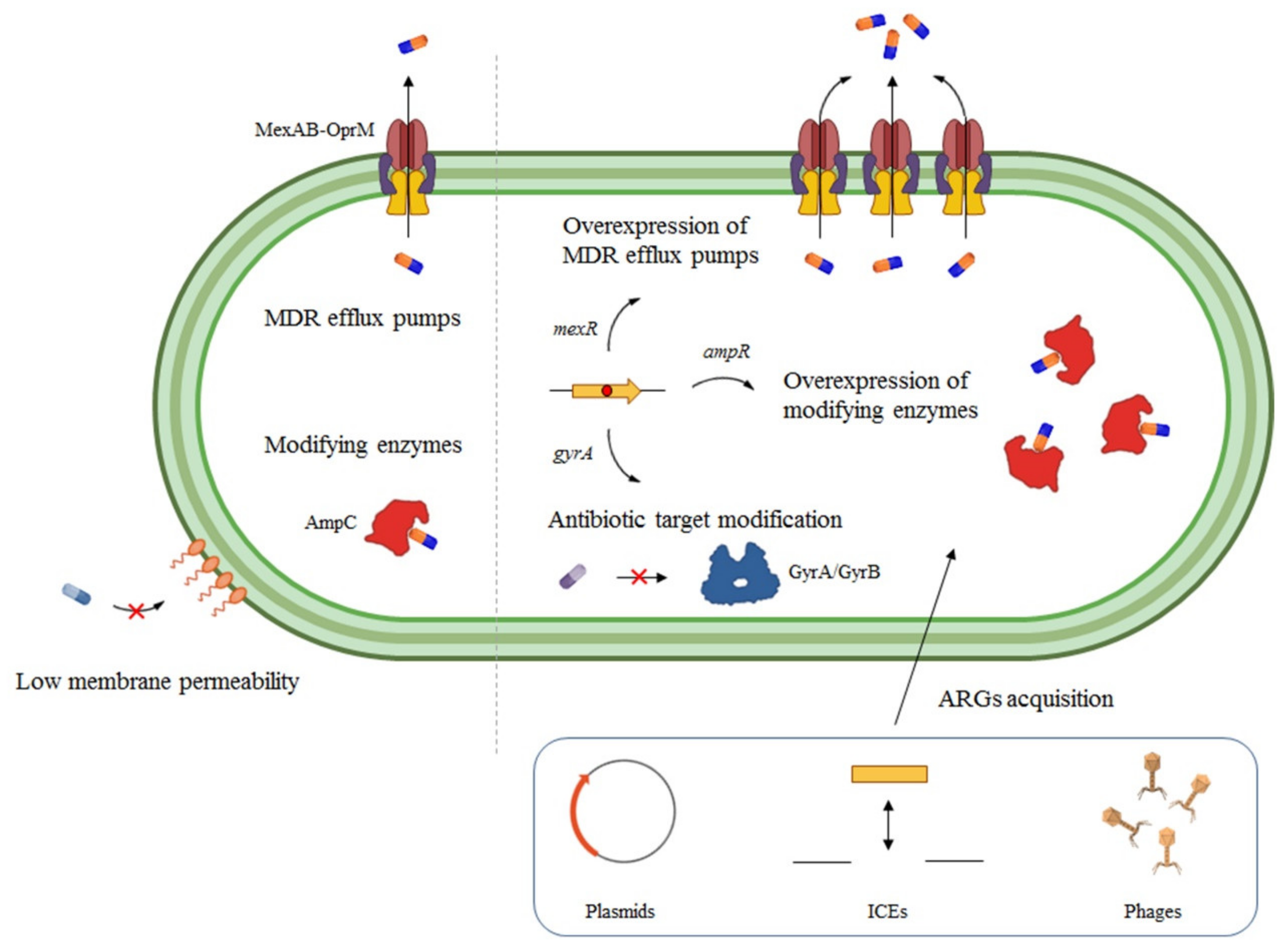
Ijms Free Full Text Coming From The Wild Multidrug Resistant Opportunistic Pathogens Presenting A Primary Not Human Linked Environmental Habitat Html
Safety If using a large can do not continue heating the can after inserting the rubber stopper as pressure will increase.
A tin is shrinking after boiled boyles law. This law is otherwise known as BoyleMariotte law or Pressure-Volume Law. Use the kitchen tongs to grab the can. Adjust the positions of the loops until the rod is horizontal.
They can occur anywhere but most often in hairy areas that sweat or rub such as. Cover a burning candle with a pitcher so that the candle is in an air-tight room sealed by the water at the ground. 20 min to boil 1 or 2 min to collapse.
The end of the rod supporting that paper bag rises. It is a homogeneous mixture made up of water and various salts including sodium chloride or common table salt. Water does so dramatically.
The pulse rate systolic blood pressure circulation rate and cardiac output increase. Apply a hot compress for 10 minutes or so three times daily to encourage the boil to come to a head. This is the reason why astronauts wear specially designed spacesuits.
In this rising water experiment you got to see firsthand the effects of water. Boyles law equation is written as. So when a full and sealed container cools the water retracts in size.
Heat the can until the water inside starts to boil. In case the spacesuit of the astronaut gets ruptured the blood and the body fluids start to boil and the astronaut gets seriously injured. Just before the candle dies the water level rises to almost 110 th of pitcher height.
After some time the candle dims and goes out. The water stops rising when that pressure equalization is reached. No air bubbles are seen.
2 Fill the bowl with cold water the colder the better. This alloy is made up of a mixture of zinc copper and nickel and it looks a lot like silver. P 1 V 1 P 2 V 2.
For example a balloon will maintain its size as long as the gas pressure inside the balloon is the same as the atmospheric pressure outside the balloon. Use Boyles Law to answer the following questions. Most all items swell somewhat with heat applied.
Presumably this is because they are coming into contact with cooler water. If one gas is removed water vapor condensing the total pressure of the gases in the container declines. Where V is the volume of the gas P is the pressure of the gas and K 1 is the constant.
Boils are painful red pus-filled lumps in your skin caused by an infection of hair follicles. The boiling point of nitrogen is -1958 degrees Celsius 774K. 4 Place the can directly on the burner of the stove while it is in the OFF position.
Leave the balance to stand without heating a bag. What is the new pressure of the gas. -1830 degrees Celsius 902K.
Heat the air below one paper bag. After 10 to 15 minutes impaired efficiency is obvious. Start by rinsing out the soda cans to remove any leftover soda goo.
Gas Law IMF Collapsing Metal Can Boyles Law Description. As the kettle starts to boil at around 255 we can see large steam bubbles being formed at the bottom where the heating element is and these bubbles shrink as they rise. Use fresh towels every time you wash and dry the infected areas.
Since the amount of water inside the can is small to hear water bubbling you can tell that it is boiling when you notice steam coming out of the can. As gases decompress their volume expands. Breathing at depth ascending and holding your breath may cause serious lung over-expansion resulting in arterial gas embolism etc.
Wash your hands thoroughly to prevent the spread of infection. PV k 1. Steinbergs richly textured Jewish family narrative.
At this point the can will. Empty soda can Beaker Ice water Bunsen burner Procedure. Taking a 1 cubic foot balloon down in the ocean would cause it to shrink to 12 its size at 33.
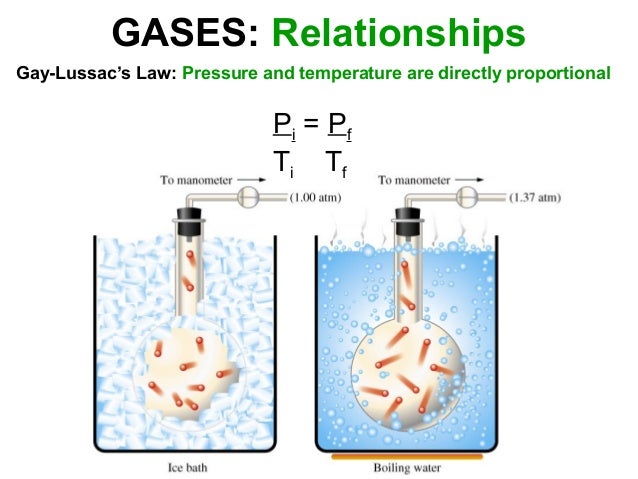
A metal can is crushed when immersed in cold water. Collapsing will take longer if the can is left to heat longer and it itself gets hot. Demonstration of Boyles Law Charless Law According to Boyles law the volume of a gas depends on the pressure it is under.
The outside air pushes water into the container until the pressure is equalized both inside and outside of the container an amazing visual on water pressure equalization. An 1810mL sample of. Heat the air below the other bag.
Boyles Law can be used to determine the current pressure or volume of gas and can be represented also as. Quickly invert the can and submerge it into the beaker filled with ice. Robert Boyle put Boyles Law into words in 1662 stating that the pressure of a gas is inversely proportional to volume of a gas.
Then we get a crazy convection current for a bit before the element switches off again. To keep it simple if there is less volume of gas then there is more pressure on the gas at constant temperature. The other end of the rod rises.
The rod becomes horizontal again. 1 100 L of a gas at standard temperature and pressure is compressed to 473 mL. The air in the balloon is primarily a mixture of nitrogen and oxygen bp.
Be careful the can is very hot so use tongs to not get burned. Respiration increases in depth and sometimes in rate. Cover a burst boil with a bandaid.
2 In a thermonuclear device the pressure of 0050 liters of gas within the bomb casing reaches 40 x 106 atm. 3 Add 1 generous tablespoon of water to the empty soda can just enough to cover the bottom of the can. As gases get compressed their volume shrinks.
At 12000 to 15000 feet however the effects of hypoxia on the nervous system become increasingly apparent. As the container collapses the volume of the container decreases. Add 15 mL of water to aluminum can and bring to a vigorous boil.
See your doctor if the boil isnt improving after a few days. The water level stays up for many few minutes more. Pressure in a container is the sum of the partial pressures of the constituent gases.
One infected hair follicle is a furuncle and a group of infected follicles joined together is a carbuncle. These absolute temperatures are between one third and one quarter of room temperature so according to Charles Law one would expect the balloon to shrink by about the same proportion. Weights or books each should be the same size Safety Wear safety goggles in.
As soon as I finished Janice Steinbergs new novel The Tin Horse Random House 26 I gave a copy to my 100-year-old Grandma Bea. Requires a large hotplate. Boyles Law Materials Required 140-mL syringe with end cap Ring stand with clamp or wood block with hole drilled in it or other device to hold syringe in an upright position.

7 Essential Oils For Itchy Bug Bites
Comments
Post a Comment