Tin Oxide Reaction With Carbon
GALVANIC REACTION CHART Below is a galvanic reaction chart for dissimilar metals. In turn carbon is oxidized to carbon monoxide and carbon dioxide.

Raman Spectra Of The Tin Oxide Thin Films Oxidized At Different Download Scientific Diagram
With many other names this oxide of tin is an important material in tin chemistry.
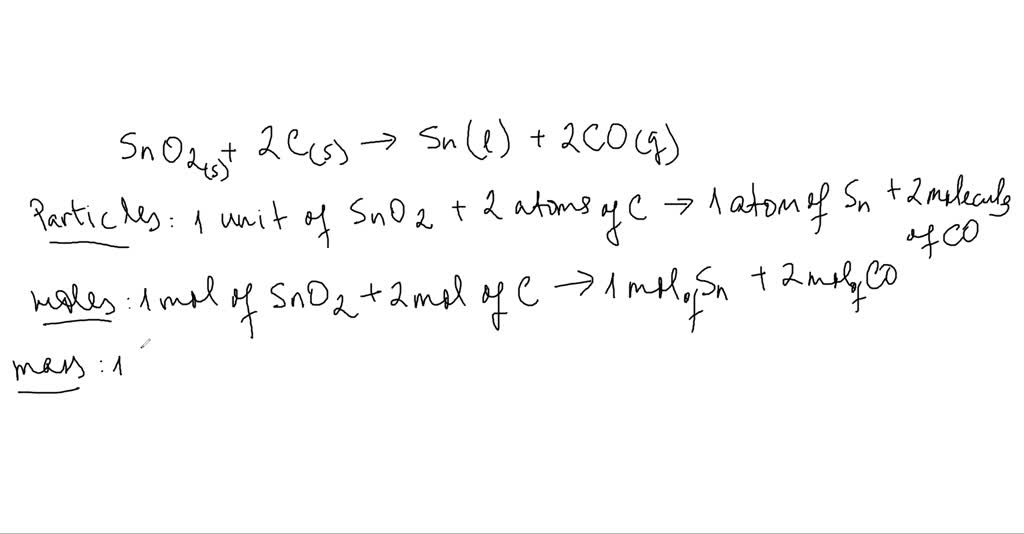
Tin oxide reaction with carbon. Carbon and silicon oxides. Where n moles SnO₂ m mass SnO₂ 140 g and M molar mass SnO₂ 11871gmol Sn 215999gmol O 15071 g SnO₂mol SnO₂ n mM ggmol g molg nSnO₂. OXIDATION OF CARBON MONOXIDE ON TINlv OXIDE 445 nrg-1.
And again calculate the entropy in the entropy. Please understand that green represents lower risk not no risk It should be noted that if sacrificial plating is incorporated in the fastener design then galvanic action can result in the deterioration of the sacrificial coating rather than of the fastener. Water content 35 are dis- cussed later.
It is normally counted as a neutral oxide. TinIV oxide also known as stannic oxide is the inorganic compound with the formula SnO 2The mineral form of SnO 2 is called cassiterite and this is the main ore of tin. It is a colourless diamagnetic amphoteric solid.
Start with a balanced equation. It doesnt react with water but it will react with hot concentrated sodium hydroxide solution to give a solution of sodium methanoate. So now what we can look at is the other reaction.
The solids may be dispensed in plastic weighing dishes. The mixture turns brown as copper is formed. So now we have slightly different units for entropy and entropy.
We proceeded to convert the less anode-active tin oxidecarbon SnO 2 C composites into the highly anode-active tin sulfidecarbon SnS 2 C materials by confined annealing with sulfur powder. Continuing on with thermodynamics we are continuing to take a look at some basic parameters with thermodynamics. If the carbon is more reactive than the metal it will remove the oxygen from the metal oxide and leave traces of the metal in the reaction vessel.
If you heat a mixture in a test tube a red glow runs through the mixture showing that heat is being produced in the reaction in addition ot the heat from the Bunsen. From a reaction enthalpy analysis we reveal that the sulfidation reaction with sulfur is possible for a wide variety of metal oxides. When we have the formation of solid tin and carbon dioxide Is 187 2 kg joules per mole and the disorder Equal to.
The oxides of carbon. Does carbon react with metal. Chemical reactions for tin.
It is best not to issue all three solids. Thermodynamic properties of substances The solubility of the substances Periodic table of elements. Answer 1 of 3.
The reaction between carbon and copperII oxide. Traditional reactive gas O 2 was replaced by CO 2 to fabricate carbon-doped tin oxide SnO x-C using pulse direct current magnetron sputtering pDC-MS with different ArCO 2 flow ratios the physical and electrochemical properties of SnO x-C films were characterized by X-ray reflection XRR X-ray photoelectron spectroscopy XPS battery tester et al. For tin oxide the reduction with carbon or coke is an established industrial process.
The dioxides of germanium tin and lead tend to have more acidic character than the monoxides. Sn s O 2 g SnO 2 s. Carbon and silicon oxides.
Carbon powder and copperII oxide are both black. However for some metals this fails and metal carbides are formed instead. Were looking at the entropy of the entropy t.
When heated tin reacts with oxygen O 2 forming tin dioxide SnO 2. This reaction takes place at a temperature of 800-900C. TinIV oxide react with carbon to produce tin and carbon monoxide.
This means that some metals can be extracted from their metal oxides using carbon. Students try leadII oxide first before using a slightly modified technique to try copperII oxide. The synthesized ATO nanoparticles had a.
This works for zinc iron tin lead and. Antimony doped tin oxide ATO and its composites with carbon nanotubes CNTs were synthesized and investigated as alternative Pt catalyst supports for the oxygen reduction reaction ORR and the methanol oxidation reaction MOR. Due to this fact it can be stated that the reactions occurring with the contribution of examined organic compounds on SnO 2 surface are multi-stage but follow the same scheme.
SnO₂ 2C 2CO Sn Calculate the moles n of SnO₂ in 140 g. This means that the oxidation reaction rates of the compounds at the surface of tin dioxide are affected to the same degree by the increase in concentration of each of the assayed compounds. Carbon dioxide is an acidic oxide a typical property of the majority of non-metal oxides and reacts with sodium hydroxide to form a salt and water.
Metal oxide carbon metal carbon dioxide. The germanium tin and lead oxides are all amphoteric. Less reactive metals than carbon can be extracted from their oxides by heating with carbon.
Under normal conditions tin does not react with air. Carbon monoxide is usually treated as if it was a neutral oxide but in fact it is very very slightly acidic. It doesnt react with water but it will react with hot concentrated sodium hydroxide solution to give a solution of sodium methanoate.
Arrhenius plots for CO oxidation on SnOa gel calcined at 450 for small tem- perature ranges between 100 and 230C showed that the apparent activation energy for the reaction increased approximately linearly with temperature giving values of 8 and 19 kcal mole-1 at 130 and 210. The electrical properties of tiniv oxide render it an excellent material for the detection of very low levels of carbon monoxide and several devices based on sintered pellets of SnO2 are. Carbon is a non-metal but it is more reactive than some metals.
The most prominent example is the production of iron from iron oxide in a blast furnace. Carbon monoxide is usually treated as if it was a neutral oxide but in fact it is very very slightly acidic. SnO2 C Sn CO Calculate the number of moles of tin that are produced if all 00949 moles of tin IV oxide are consumed during the reaction.
00949 moles of tin IV oxide reacts with carbon according to the reaction equation shown below. Carbon monoxide is only slightly soluble in water and doesnt react with it.
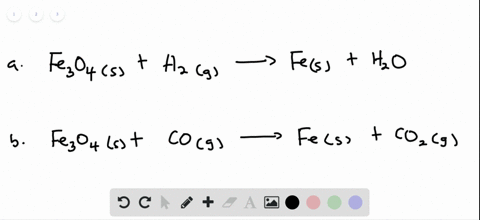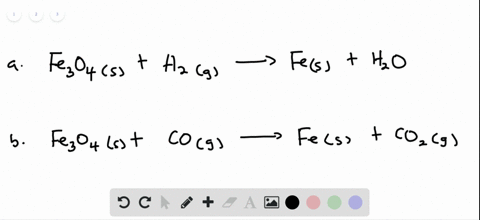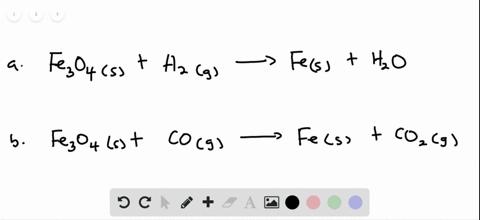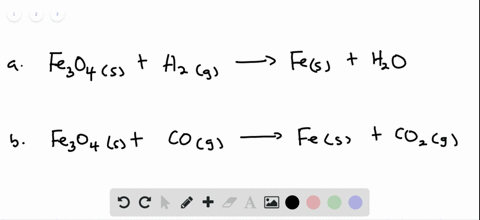
Solved Smelting When Tin Iv Oxide Is Heated With Carbon In A Process Called Smelting The Element Tin Can Be Extracted Begin Equation Mathrm Sno 2 Mathrm S 2 Mathrm C Mathrm S Rightarrow Mathrm Sn 1 2 Mathrm Co Mathrm G End

In Situ Growth Of Ag Reduced Graphene Oxide Carbon Nanotube On Indium Tin Oxide And Its Application For Electrochemical Sensing Sciencedirect
Comments
Post a Comment